Otoimplant
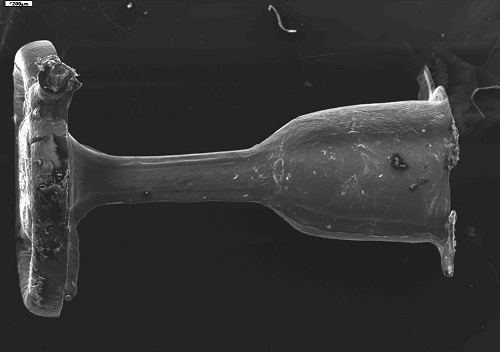
The assumption of presented project is development of middle ear prosthesis made of medical, biostable, thermoplastic polymers with bactericidal properties thanks to additions of nanosilver particles. Described in the project, as well as shown in patent application named: „Formation method of a middle ear prosthesis” WO2013/184010A3. Proposed implant will allow reconstruction of ossicular chain and will significantly decrease risk of bacterial infection. Proposed prosthesis may be treated as multifunctional implant with bacteriadical properties, will shorten necessary hospitalization, reduce risk of complication after infection, will allow proper recovery of bone structure and reinstate proper resonance of ossicular chain. Proposed implant may be applicable to patients with hearing loss related to damage of ossicular chain caused by inflammations, injuries, congential defects and otosclerosis.
The main aim of the project is to develop and evaluate usability of bactericidal prosthesis so the implants could be used in healthcare. Implant will be made of composites, biostable thermoplastic polymers and silver nanoparticles. Implants will be obtain by extrusion and injection moulding of polymer and composite materials. All necessary research that is: physicochemical and biological assessment, in vitro and in vivo (on animals) and histopathological, laryngological and audiological (clinical tests) have been included in project schedule.
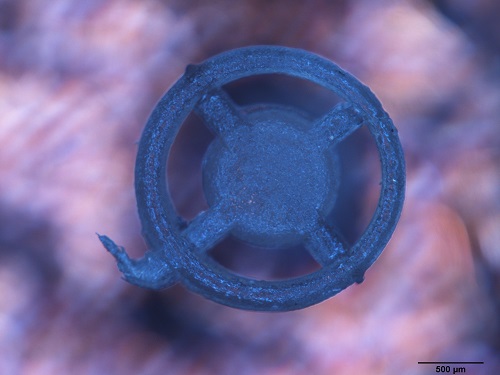
Project stage: one. Assessment of physiochemical and structural properties (SEM + EDS), X-ray diffraction (XRD) and DSC, DTA/Tg researches. Basic mechanical and thermal parameters will be rated, surface characterization according to roughness and wettability. Kinetics of silver ion Ag+ release will be tested in in vitro using ICP-MS method. Prosthesis will be tested paying attention to their cytotoxicity and bacteriadical properties. Above will be commited using fibroblasts and osteoblasts cell lines. Bacteriadical effectiveness researche will be performed on Gram-positive and Gram-negative bacteria. Research outcome will determine implants showing best bacteriadical properties and will also be key factor allowing proceeding to second stage.
Project stage: two. Assessment do biological properties of prosthesis in in vivo on rats. Planned research methods: histological, histochemical, histoenzimatical. Research outcome will determine implants showing the greatest success factor and will decide about moving to third stage.
Project stage: three. The most important stage of the project including clinical tests on humans. Thanks to cooperation with Department of Otolaryngology of University Hospital in Kraków and in response to market demand for reconstruction of ossicular chain using implants it will be possible to commit pilot programme of surgeries. New hospital procedures will be prepared to guarantee the highest level of medical service. It’s planned to commit to surgeries on every patient. During first one, implant will be placed in middle ear, patient will be hospitalized and further examinations will be committed: otolaryngological, audiological, tone audiograms, speech audiograms and impedance audiometry. Above examinations will be performed in certain time intervals. After all second surgery will be necessary to examine tissues around implanted prosthesis. Project will finish with summary and outcome elaboration what will help in future commercialization process.

As the project outcome we will consider collection of complex research effects gained by research team. If the project outcome will be as expected by the project assumptions, new method of surgery in otolaryngology will be delivered. Designed prosthesis made of biostable thermoplastic polymers with addition of silver nanoparticles will be used during common surgery. Implantation of newly designed prosthesis will recover ossicular chain functionalities and in consequence will enhance sense of hearing. Proposed implant might be successfully used in present surgery. Positive results of committed researches will allow wide group of patients to get access to modern medical product. Cheaper, more accessible implants with bactericidal properties will support antibiotic treatment. Developed prosthesis will act like a barrier protecting from reoccurring illness and inflammation what will shorten necessary hospitalization and accelerate convalescence.
